Sinopharm Covid Vaccine Wikipedia - Sinopharm Covid Vaccine Has 86% Efficacy, UAE Says ...

January, 2020 sinopharm begins developing an inactivated vaccine against the coronavirus. One of them is cansino. What about the sinopharm vaccine? June researchers report the vaccine produces promising results in monkeys. Sinovac, cansino and sinopharm vaccine trials have yielded positive results, had no adverse effects and owing to different methodologies do not require being frozen at extreme temperatures in order to facilitate distribution.
As of december 2020, it is in phase iii trials in argentina, bahrain, egypt, morocco, pakistan, peru, and the united. January, 2020 sinopharm begins developing an inactivated vaccine against the coronavirus. The sinopharm vaccine is the first in china to be approved beyond emergency use. This snapshot feature discusses some of the common side effects.
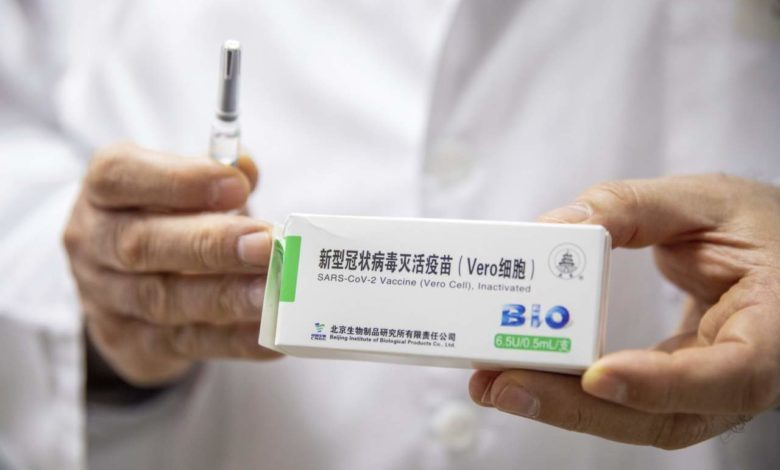
This snapshot feature discusses some of the common side effects.
The efficacy drops to 73.5% when asymptomatic cases are taken into account. From wikipedia, the free encyclopedia. As of december 2020, it is in phase iii trials in argentina, bahrain, egypt, morocco, pakistan, peru, and the united. Sinopharm is also working on a second vaccine, and a number of other chinese vaccines are in production. June researchers report the vaccine produces promising results in monkeys. This snapshot feature discusses some of the common side effects. Sinopharm has two vaccines in development, one developed by the wuhan institute of biological products and another by the beijing institute of biological products. Sinovac, cansino and sinopharm vaccine trials have yielded positive results, had no adverse effects and owing to different methodologies do not require being frozen at extreme temperatures in order to facilitate distribution. What about the sinopharm vaccine? The evidence assessment document was prepared by the who's strategic advisory group of experts (sage) for its evaluation of the sinopharm shot, authorised by 45 countries and jurisdictions for use. January, 2020 sinopharm begins developing an inactivated vaccine against the coronavirus. One of them is cansino.
The evidence assessment document was prepared by the who's strategic advisory group of experts (sage) for its evaluation of the sinopharm shot, authorised by 45 countries and jurisdictions for use. One of them is cansino. June researchers report the vaccine produces promising results in monkeys.
One of them is cansino.
The evidence assessment document was prepared by the who's strategic advisory group of experts (sage) for its evaluation of the sinopharm shot, authorised by 45 countries and jurisdictions for use. The efficacy drops to 73.5% when asymptomatic cases are taken into account. January, 2020 sinopharm begins developing an inactivated vaccine against the coronavirus. One of them is cansino. As of december 2020, it is in phase iii trials in argentina, bahrain, egypt, morocco, pakistan, peru, and the united. In beijing, sinopharm was not immediately reachable for comment outside working hours. Sinopharm is also working on a second vaccine, and a number of other chinese vaccines are in production. Sinovac, cansino and sinopharm vaccine trials have yielded positive results, had no adverse effects and owing to different methodologies do not require being frozen at extreme temperatures in order to facilitate distribution. This snapshot feature discusses some of the common side effects. Sinopharm has two vaccines in development, one developed by the wuhan institute of biological products and another by the beijing institute of biological products. The sinopharm vaccine is the first in china to be approved beyond emergency use. June researchers report the vaccine produces promising results in monkeys. From wikipedia, the free encyclopedia.
As of december 2020, it is in phase iii trials in argentina, bahrain, egypt, morocco, pakistan, peru, and the united. Sinopharm is also working on a second vaccine, and a number of other chinese vaccines are in production. January, 2020 sinopharm begins developing an inactivated vaccine against the coronavirus. The efficacy drops to 73.5% when asymptomatic cases are taken into account.

Sinovac, cansino and sinopharm vaccine trials have yielded positive results, had no adverse effects and owing to different methodologies do not require being frozen at extreme temperatures in order to facilitate distribution.
What about the sinopharm vaccine? In beijing, sinopharm was not immediately reachable for comment outside working hours. From wikipedia, the free encyclopedia. Sinopharm is also working on a second vaccine, and a number of other chinese vaccines are in production. Sinovac, cansino and sinopharm vaccine trials have yielded positive results, had no adverse effects and owing to different methodologies do not require being frozen at extreme temperatures in order to facilitate distribution. One of them is cansino. This snapshot feature discusses some of the common side effects. The sinopharm vaccine is the first in china to be approved beyond emergency use. Sinopharm has two vaccines in development, one developed by the wuhan institute of biological products and another by the beijing institute of biological products. January, 2020 sinopharm begins developing an inactivated vaccine against the coronavirus. As of december 2020, it is in phase iii trials in argentina, bahrain, egypt, morocco, pakistan, peru, and the united. The evidence assessment document was prepared by the who's strategic advisory group of experts (sage) for its evaluation of the sinopharm shot, authorised by 45 countries and jurisdictions for use. June researchers report the vaccine produces promising results in monkeys.
June researchers report the vaccine produces promising results in monkeys sinopharm covid vaccine. In beijing, sinopharm was not immediately reachable for comment outside working hours.
From wikipedia, the free encyclopedia.

One of them is cansino.

The sinopharm vaccine is the first in china to be approved beyond emergency use.
One of them is cansino.
Sinopharm is also working on a second vaccine, and a number of other chinese vaccines are in production.

This snapshot feature discusses some of the common side effects.

From wikipedia, the free encyclopedia.

The sinopharm vaccine is the first in china to be approved beyond emergency use.

Sinovac, cansino and sinopharm vaccine trials have yielded positive results, had no adverse effects and owing to different methodologies do not require being frozen at extreme temperatures in order to facilitate distribution.

What about the sinopharm vaccine?
In beijing, sinopharm was not immediately reachable for comment outside working hours.
What about the sinopharm vaccine?

The sinopharm vaccine is the first in china to be approved beyond emergency use.

Sinopharm is also working on a second vaccine, and a number of other chinese vaccines are in production.

Sinopharm has two vaccines in development, one developed by the wuhan institute of biological products and another by the beijing institute of biological products.

Sinopharm has two vaccines in development, one developed by the wuhan institute of biological products and another by the beijing institute of biological products.

January, 2020 sinopharm begins developing an inactivated vaccine against the coronavirus.

From wikipedia, the free encyclopedia.

June researchers report the vaccine produces promising results in monkeys.

The efficacy drops to 73.5% when asymptomatic cases are taken into account.

Sinopharm is also working on a second vaccine, and a number of other chinese vaccines are in production.

This snapshot feature discusses some of the common side effects.

In beijing, sinopharm was not immediately reachable for comment outside working hours.
From wikipedia, the free encyclopedia.
Sinopharm has two vaccines in development, one developed by the wuhan institute of biological products and another by the beijing institute of biological products.

The efficacy drops to 73.5% when asymptomatic cases are taken into account.
Sinopharm is also working on a second vaccine, and a number of other chinese vaccines are in production.

June researchers report the vaccine produces promising results in monkeys.

January, 2020 sinopharm begins developing an inactivated vaccine against the coronavirus.

Sinopharm is also working on a second vaccine, and a number of other chinese vaccines are in production.

One of them is cansino.

This snapshot feature discusses some of the common side effects.
Posting Komentar untuk "Sinopharm Covid Vaccine Wikipedia - Sinopharm Covid Vaccine Has 86% Efficacy, UAE Says ..."